Unit 5 – Lipids: Fats, Oils, and Cholesterol
5.3 Three Classes of Lipids
Lipids are the family of organic nutrients that are mostly insoluble in water, meaning they do not mix well with water. The three main types of lipids are triglycerides, phospholipids, and sterols. On this page, we’ll learn about the structures of these three, as well as their functions in the body, and their content in various foods. Before reading on, watch the 4:07 minute TED Ed video “What if Fat?” that introduces the types of fatty acids.
VIDEO: “What is Fat?” By George Zaidan for TED Ed. (4:07 minutes) May 22, 2013
Triglycerides
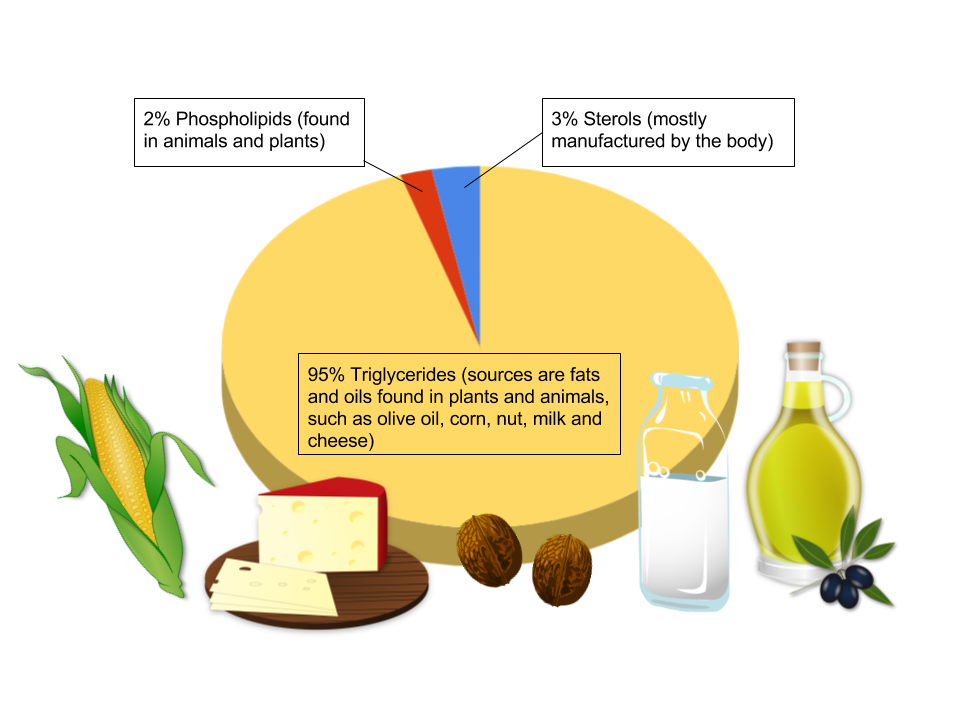
Triglycerides are the main form of lipids in the body and in foods. More than 95 percent of lipids in the diet are in the form of triglycerides. Visible fats are those you can rediliy see such as butter, vegetable oil, and the marbling of fat in meat. Hidden fats are those in baked goods, dairy products like milk and cheese, and fried foods. We commonly call the triglycerides that are solid “fats” and those that are liquid are “oils.” The terms fats, oils, and triglycerides are often used interchangeably. In this unit, when we use the word “fat,” we are referring to triglycerides.
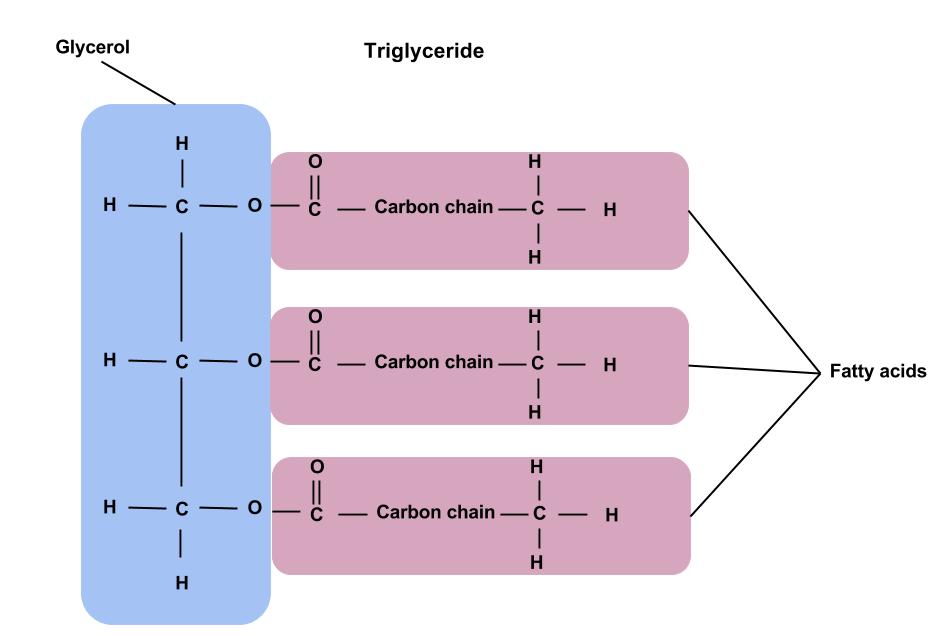
A triglyceride is composed of a three-carbon glycerol backbone to which three fatty acids are attached. The “glyceride” in the word “triglyceride” refers to this glycerol backbone, while the “tri” refers to the fact that there are three fatty acids attached. A monoglyceride contains glycerol with one fatty acid attached, and a diglyceride contains glycerol with two fatty acids attached.
Triglycerides generally are composed of a mixture of different types of fatty acids. Foods contain different proportions of fatty acid types, and this influences disease risks associated with dietary patterns. We will take a closer look at these differences, along with food sources, in the next section.
Phospholipids
Phospholipids are found in both plants and animals but make up only about 2 percent of dietary lipids. However, they play many important roles in the body and in foods. Phospholipids can also be synthesized by the body, so they don’t have to be consumed in the diet.
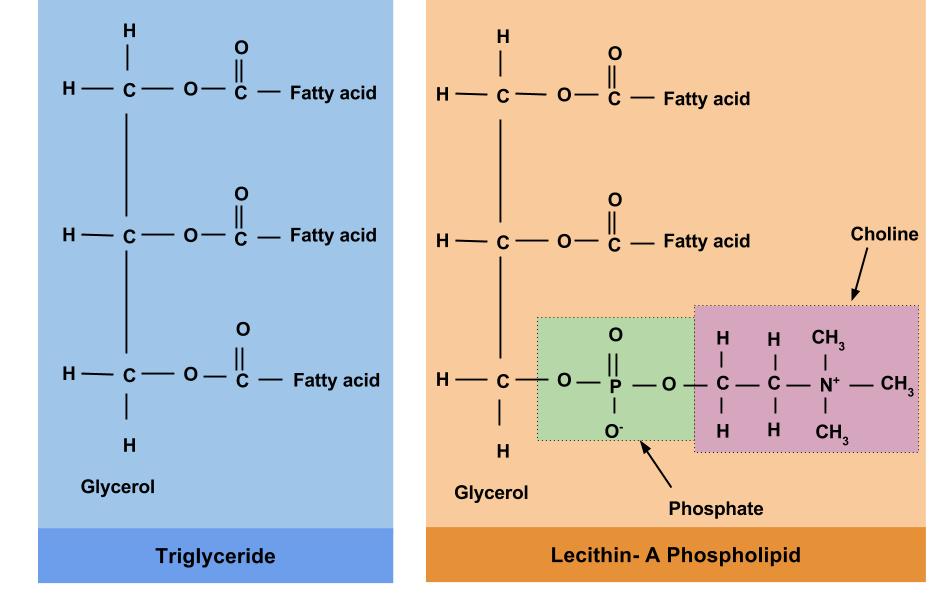
Phospholipids are similar in structure to triglycerides (Figure 5.13). Like triglycerides, phospholipids have a glycerol backbone. But unlike triglycerides, phospholipids only have two fatty acid molecules attached to the glycerol, while the third carbon of the glycerol is bonded to a phosphate group.
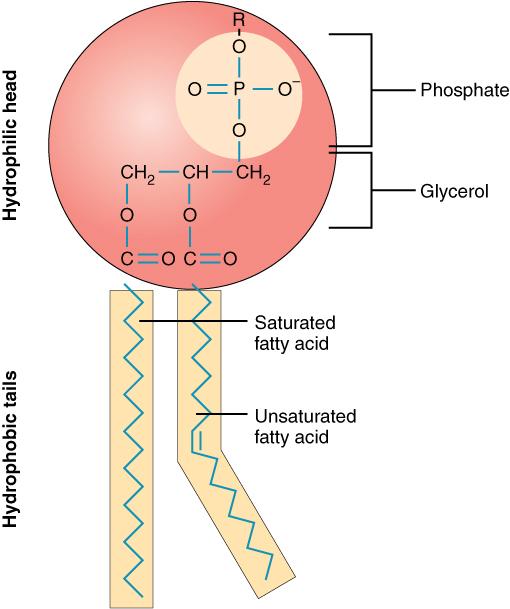
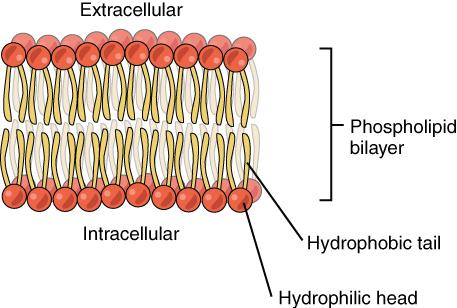
The unique structure of phospholipids makes them both fat- and water-soluble, or amphiphilic. The fatty-acids are hydrophobic (dislike water), and the phosphate group and glycerol are hydrophilic (attracted to water) as noted in the figure below.
The amphiphilic nature of phospholipids makes them very useful for several functions in the body. Every cell in the body is encased in a membrane composed primarily of a double layer of phospholipids (also known as the phospholipid bilayer) as noted in Figure 5:14. This double layer protects the inside of the cell from the hydrophobic outside environment while at the same time allowing for transport of fat and water through the membrane. Phospholipids also play a role in transporting fats in the blood, as we’ll learn later in this unit.
Phopholipids as Emulsifiers
Another important role of phospholipids is to act as emulsifiers. Emulsions are mixtures of two liquids that do not normally mix (oil and water, for example). Without an emulsifier, the oil and water separate out into two layers. Because of their ability to mix with both water and fat, phospholipids are ideal emulsifiers that can keep oil and water mixed, dispersing tiny oil droplets throughout the water. Lecithin—a phospholipid found in egg yolk, soybean, and wheat germ—is often used as a food emulsifier. Emulsifiers also play an important role in making food appetizing; their inclusion in foods like sauces and creams makes for a smoother texture and prevents the oil and water ingredients from separating out. They also can extend shelf life.
Sterols
Sterols have a very different structure than triglycerides and phospholipids. Sterols consist of multi-ring structures, similar to chicken wire. They are complex molecules that contain interlinking rings of carbon atoms, with side chains of carbon, hydrogen, and oxygen attached.
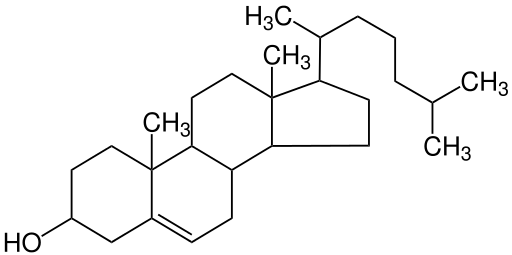
Cholesterol is the best-known sterol because of its role in heart disease. It forms a large part of the fatty plaques that narrow arteries and obstruct blood flow in atherosclerosis. However, cholesterol also has many essential functions in the body. Like phospholipids, cholesterol is present in all body cells as it is an important substance in cell membrane structure. Cholesterol is also used in the body as a precursor in the synthesis of a number of important substances, including vitamin D, bile, and sex hormones such as progesterone, testosterone, and estrogens.
Cholesterol is not an essential nutrient; it does not need to be consumed in the diet, because it is manufactured in the liver. Only foods that come from animal sources contain cholesterol. Cholesterol is found in foods like meat, poultry, fish, egg yolks, butter, and dairy products made from whole milk.
Plant foods do not contain cholesterol, but sterols found in plants resemble cholesterol in structure. Plant sterols inhibit cholesterol absorption in the body, which can contribute to lower cholesterol levels. Plant sterols occur naturally in vegetable oils, nuts, seeds, and whole grains. In addition, some foods like margarines and dressings are fortified with plant sterols.
Review Questions
Attributions:
- Lane Community College’s Nutrition: Science and Everyday Application “Classification of Nutrients ” CC BY-NC 4.0
Image Credits:
- Figure 5.5. “Types of Fat” by Allison Calabrese is licensed under CC BY 4.0
- Figure 5.6. “The Structure of a Triglyceride” by Allison Calabrese is licensed under CC BY 4.0
- Figure 5.7. “Simple Triglyceride Diagram” by Alice Calahan is licensed under CC BY-SA 4.0
- Figure 5.8. “The Difference Between Triglycerides and Phospholipids” by Allison Calabrese is licensed under CC BY 4.0
- Figure 5.9. “Phospholipid Structure” by J. Gordon Betts, Kelly A. Young, James A. Wise, Eddie Johnson, Brandon Poe, Dean H. Kruse, Oksana Korol, Jody E. Johnson, Mark Womble, Peter DeSaix is licensed under CC BY 4.0
- Figure 5.10. “Phospolipid Bilayer” by J. Gordon Betts, Kelly A. Young, James A. Wise, Eddie Johnson, Brandon Poe, Dean H. Kruse, Oksana Korol, Jody E. Johnson, Mark Womble, Peter DeSaix is licensed under CC BY 4.0
- Figure 5.11. “Cholesterol Chemical Structure” by Wesalius is in the Public Domain
In chemistry and nutrition, organic means carbon containing.
The main type of lipid in the body and food supply. Concist of a glycerol backbone with three fatty acids attached.
Lipids composed of a glycerol backbone with two fatty acids and one phosphorus containing group attached.
The group of lipid with a ring sturcture. Examples include cholesterol, estrogen, and tesostorone.
Being both hydrophilic and hydrophobic--tit attracts both fats and water
Repels water.

